- Blog
- Microsoft office word viewer free download
- New mexico time zone map
- Isosceles and equilateral triangles worksheet answers
- Powerpoint free economic infographics templates
- Alphabetical recycling spelling words
- Google map philippines street view satellite
- Moca cognitive assessment pdf
- Online pdf editor free download
- Convert gif to webm adobe media encoder
- Reasons for leaving a job on application
- 1st studio siberian mouse masha babko
- Mmpi free online test reddit
- Fairway solitaire online free
- Skyrim unlock all alchemy recipes mod
- Building brochure design templates free download
- Roadblock app game
- Sample personal budget items
- Xbox 360 iso downloads emulator
- Volleyball signals for referee
- Spectrum basic tv service channel list
- Acer aspire laptop windows 7 driver downloads
- Switching channels spectrum tv choice
- Was sonic mania a fan game
- The density of water
- Usps scheduling for passports
- Windows time clock app free
- Banktivity 8 user manual pdf
- Spectrum tv cable watch live
- Calorie and macro tracker spreadsheet template
- Used mini excavator buckets for sale
- Arduino mega isp pinout
- Printable checkbook register free
- Eagle eye exterminating
- Density of water gml
- Genogram social work software for pc
- Among us mobile logo
- Sweets Bakery food Baked blender donuts
- Aarm for traxxas slash ultimate
- Meal planning blank sheets
- Wife swaping hot urdu sex story
- John dalton atomic theory date
- Iso 13485 internal audit checklist free
- Cheese cake stater bros
- Logitech gaming software not detecting g29
- Prolific pl2303 windows 10 driver error
- Manycam free download old version with zoom
- Foxit phantompdf educational activation key free
- Back of neck lymph nodes
- Ps2 initial d usa iso
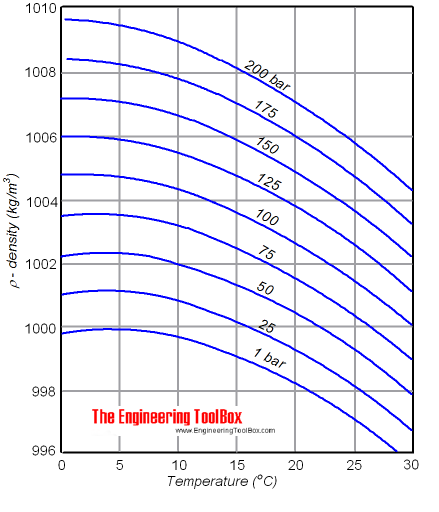
Therefore, the ρ solid > ρ liquid > ρ gas Solid atoms or molecules are more closely packed together than liquid atoms or molecules. In general, a solid mass of a substance sinks when immersed in a liquid of the same substance. The less-dense stuff floats on top of the dense liquid.īecause the ρwater of ice is smaller than the ρwater of water, ice floats on water. The density of water (g/ml) = 8kg/(8 x10-3 m3 ) The mass (m) of 8 liters of water is 8 kg. Specific weight (γ) = (ρ x volume x g)/volume It is represented by the symbol γ.Īt 4☌, the specific weight of water on earth is 9.807 kN/m 3 = 62.43 lbs/ft 3.Ĭheck the full article “Specific gravity of water”. The specific weight of water is the weight of water per unit volume. There are three different Units of Gallon: Weight of a Gallon of Water Weight of 1 liter (l) of pure water (at 4 ☌) = 1 kilogram (kg). The weight of a gallon of water depends upon storing the temperature of the water, For the sake of the question, ‘How much does a gallon of water weigh?’, we are going to focus on the weight of water at 62 ☏ (17 ☌).ġ US gal of water = 8.345 pounds = 3.785 kg (at 17 ☌).ġ Imperial gal of water = 10.02 pounds = 4.545 kg (at 17 ☌). At 4☌, the density of fresh water on Earth is generally understood to be 1000 kg/m 3. However, this value varies depending on the temperature and impurities in the water.Ĭommon units of ρ water are grams per milliliter (1 g/ml), grams per cubic centimeter ( g/cm 3), and pounds per cubic foot (lb/ft 3). The density of water (ρ water) is around 8.345 lbs/U.S. This is since iron has a greater amount of substance than wood.ĭensity (ρ) formula = mass (m) of substance ÷ volume (v) of substance The density of water in lbs/U.S. We’ll find that iron is heavier than wood.

It is the mass-to-volume ratio of the material. How much does a gallon of water weigh at its highest density?ĭensity refers to the amount of substance per unit volume.The density of Water in g/ml (Effect of Temperature).Calculating the specific weight of water.
- Blog
- Microsoft office word viewer free download
- New mexico time zone map
- Isosceles and equilateral triangles worksheet answers
- Powerpoint free economic infographics templates
- Alphabetical recycling spelling words
- Google map philippines street view satellite
- Moca cognitive assessment pdf
- Online pdf editor free download
- Convert gif to webm adobe media encoder
- Reasons for leaving a job on application
- 1st studio siberian mouse masha babko
- Mmpi free online test reddit
- Fairway solitaire online free
- Skyrim unlock all alchemy recipes mod
- Building brochure design templates free download
- Roadblock app game
- Sample personal budget items
- Xbox 360 iso downloads emulator
- Volleyball signals for referee
- Spectrum basic tv service channel list
- Acer aspire laptop windows 7 driver downloads
- Switching channels spectrum tv choice
- Was sonic mania a fan game
- The density of water
- Usps scheduling for passports
- Windows time clock app free
- Banktivity 8 user manual pdf
- Spectrum tv cable watch live
- Calorie and macro tracker spreadsheet template
- Used mini excavator buckets for sale
- Arduino mega isp pinout
- Printable checkbook register free
- Eagle eye exterminating
- Density of water gml
- Genogram social work software for pc
- Among us mobile logo
- Sweets Bakery food Baked blender donuts
- Aarm for traxxas slash ultimate
- Meal planning blank sheets
- Wife swaping hot urdu sex story
- John dalton atomic theory date
- Iso 13485 internal audit checklist free
- Cheese cake stater bros
- Logitech gaming software not detecting g29
- Prolific pl2303 windows 10 driver error
- Manycam free download old version with zoom
- Foxit phantompdf educational activation key free
- Back of neck lymph nodes
- Ps2 initial d usa iso
