- Blog
- Microsoft office word viewer free download
- New mexico time zone map
- Isosceles and equilateral triangles worksheet answers
- Powerpoint free economic infographics templates
- Alphabetical recycling spelling words
- Google map philippines street view satellite
- Moca cognitive assessment pdf
- Online pdf editor free download
- Convert gif to webm adobe media encoder
- Reasons for leaving a job on application
- 1st studio siberian mouse masha babko
- Mmpi free online test reddit
- Fairway solitaire online free
- Skyrim unlock all alchemy recipes mod
- Building brochure design templates free download
- Roadblock app game
- Sample personal budget items
- Xbox 360 iso downloads emulator
- Volleyball signals for referee
- Spectrum basic tv service channel list
- Acer aspire laptop windows 7 driver downloads
- Switching channels spectrum tv choice
- Was sonic mania a fan game
- The density of water
- Usps scheduling for passports
- Windows time clock app free
- Banktivity 8 user manual pdf
- Spectrum tv cable watch live
- Calorie and macro tracker spreadsheet template
- Used mini excavator buckets for sale
- Arduino mega isp pinout
- Printable checkbook register free
- Eagle eye exterminating
- Density of water gml
- Genogram social work software for pc
- Among us mobile logo
- Sweets Bakery food Baked blender donuts
- Aarm for traxxas slash ultimate
- Meal planning blank sheets
- Wife swaping hot urdu sex story
- John dalton atomic theory date
- Iso 13485 internal audit checklist free
- Cheese cake stater bros
- Logitech gaming software not detecting g29
- Prolific pl2303 windows 10 driver error
- Manycam free download old version with zoom
- Foxit phantompdf educational activation key free
- Back of neck lymph nodes
- Ps2 initial d usa iso
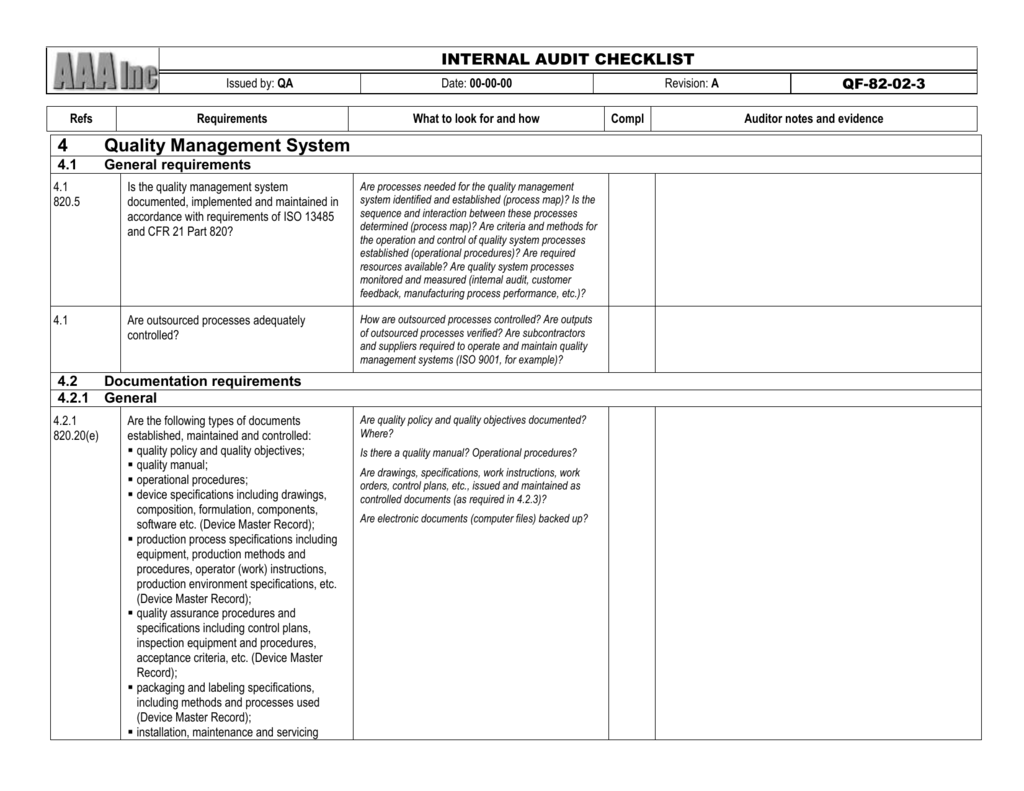
My company is small and low on the $$, and this will probably come out of my own pocket if I get it. However, I wanted to see if anyone had used it and their opinion, before paying for it. (I'd put the link in, but I haven't posted enough yet to do this).įrom the sample pages it looks great, with the right questions that will help me, as a novice, look for the right things during an audit, as well as be prepared for what external auditors will look for. ISO 13485 and FDA QSR (21 CFR 820) INTERNAL AUDIT CHECKLIST. While stumbling around the Internet looking for help I came across this: We've recently been audited and one finding was that the document we are using as a checklist/assessment tool does not cover ISO 13485, IVDD and FDA requriements (and we are subject to all of these). I've searched the forums for this topic, but couldn't find it so I apologize if it's already been discussed. ISO 134 + Additional Requirements (and missing a few ISO 9001 requirements.) This checklist adds US FDA QSR (21CFR 820) requirements (highlighted in Yellow) Here is a basic summary of the steps: Prepare your audit schedule. You don’t want to cut corners with audits – buy a complete solution! Need to train your internal auditors? Buy our Internal Audit Checklist together with Internal Auditor Training Materials and save.I am new to the forum and new to the world of Quality, so please bear with me! The checklist provides questions that refer to the ISO 13485: 2003 standard & US FDA requirements. During assessment of each requirement, auditors record the status of the evaluation by indicating in the right-hand column of the checklist a: Yes – for Acceptable Condition or No – for Deficient Condition

The bold numbers and tittles used in the first two columns of the checklist indicate the “Requirements” and may be referred to on nonconformity reports prepared by the auditor. Evidence for visible top management leadership, commitment and quality management action must be looked for. When using the checklist, the auditors must use a great deal of discretion and therefore must be careful and thoughtful prior to establishing a deficiency against a requirement. The auditors are expected to keep in mind that the AS standard does not require mandatory procedures for the various QMS processes however, the auditors will expect documented information to be available because in the clauses of the standard, the phrase such as ‘documented procedures’ is used to specify that a process, a method, a system, a work instruction, or an arrangement be documented. The additions for ISO 13485 over AS 9100 are highlighted in green and the relevant ISO 13485:2016 clause number appears with the audit question.Iso 13485 2016 Internal Audit Checklist Pdf: Fill & Download for Free GET FORM. Its huge collection of forms can save your time and jumpstart your efficiency massively. The AS 9100 Rev D standard includes the requirements of ISO 9001:2015 and additional aviation, space, and defense (ASD) industry requirements are highlighted in yellow. A free checklist tool that will help you leverage your internal quality audits for better external quality audit outcomes. CocoDoc is the best platform for you to go, offering you a free and customizable version of Iso 13485 2016 Internal Audit Checklist Pdf as you want.While both the AS 9100 and ISO 13485 standards deal with Quality Management Systems, the structures of the AS and ISO standards do not exactly line up when comparing the contents, the new requirements and / or new terminology. You will see questions on the checklist that refer to the standard and for each clause provisions are made for additional questions. The checklist is best used by trained and practicing auditors to evaluate or assess Quality Management Systems requirements based on the standards as you include the requirements of ISO 13485:2016. The internal audit checklist is based on the information provided in the 2016-09 revision of the AS 9100 Rev D, SAE standard and in the 2016 third edition of ISO 13485:2016 international standard. ISO 13485 PowerPoint Training Materials.ISO 13485 Online Training Course Listing.ISO 13485 Internal Auditor Training Courses.ISO 13485 Lead Auditor Training Courses ISO 13485 Internal Audit Checklist - View presentation slides online.Simple Steps to ISO 13485 Certification Follow our proven and manageable step-by-step process for a successful ISO implementation project. While the ISO 13485:2016 standard does not include requirements that state an internal audit checklist must be used, it is a useful and effective way to document the questions you need to ask to ensure that your process outputs meet the planned arrangements for your process.
- Blog
- Microsoft office word viewer free download
- New mexico time zone map
- Isosceles and equilateral triangles worksheet answers
- Powerpoint free economic infographics templates
- Alphabetical recycling spelling words
- Google map philippines street view satellite
- Moca cognitive assessment pdf
- Online pdf editor free download
- Convert gif to webm adobe media encoder
- Reasons for leaving a job on application
- 1st studio siberian mouse masha babko
- Mmpi free online test reddit
- Fairway solitaire online free
- Skyrim unlock all alchemy recipes mod
- Building brochure design templates free download
- Roadblock app game
- Sample personal budget items
- Xbox 360 iso downloads emulator
- Volleyball signals for referee
- Spectrum basic tv service channel list
- Acer aspire laptop windows 7 driver downloads
- Switching channels spectrum tv choice
- Was sonic mania a fan game
- The density of water
- Usps scheduling for passports
- Windows time clock app free
- Banktivity 8 user manual pdf
- Spectrum tv cable watch live
- Calorie and macro tracker spreadsheet template
- Used mini excavator buckets for sale
- Arduino mega isp pinout
- Printable checkbook register free
- Eagle eye exterminating
- Density of water gml
- Genogram social work software for pc
- Among us mobile logo
- Sweets Bakery food Baked blender donuts
- Aarm for traxxas slash ultimate
- Meal planning blank sheets
- Wife swaping hot urdu sex story
- John dalton atomic theory date
- Iso 13485 internal audit checklist free
- Cheese cake stater bros
- Logitech gaming software not detecting g29
- Prolific pl2303 windows 10 driver error
- Manycam free download old version with zoom
- Foxit phantompdf educational activation key free
- Back of neck lymph nodes
- Ps2 initial d usa iso
